EX-99.2
Published on May 5, 2026

Abdakibart (AVTX-009) in Moderate-to-Severe Hidradenitis Suppurativa Positive Topline Results from the Phase 2 LOTUS Study May 2026 | AVALO THERAPEUTICS, INC. (AVTX) 1 Exhibit 99.2

Forward-Looking Statements This presentation includes forward-looking statements made pursuant to the Private Securities Litigation Reform Act of 1995 and other federal securities laws. Forward-looking statements are statements that are not historical facts. Such forward-looking statements are subject to significant risks and uncertainties that are subject to change based on various factors (many of which are beyond our control), which could cause actual results to differ from the forward-looking statements. Such statements may include, without limitation, statements with respect to our plans, objectives, projections, expectations and intentions and other statements identified by words such as “projects,” “may,” “might,” “will,” “could,” “would,” “should,” “continue,” “seeks,” “aims,” “predicts,” “believes,” “expects,” “anticipates,” “estimates,” “intends,” “plans,” “potential,” or similar expressions (including their use in the negative), or by discussions of future matters such as: therapeutic potential, clinical benefits and safety profiles of abdakibart (AVTX-009); expectations regarding timing, success and data announcements of ongoing preclinical studies and clinical trials; the preliminary cross-study assessments comparing non-head-to-head clinical data of abdakibart to published data for lutikizumab, sonelokimab, povorcitinib, bimekizumab, secukinumab and adalimumab; integration of abdakibart into our operations; drug development costs, reliance on investigators and enrollment of patients in clinical trials; our plans to develop and commercialize our current and any future product candidates and the implementation of our business model and strategic plans for our business, current; and any future product candidates. Any forward-looking statements are based on management’s current expectations and beliefs and are subject to a number of risks, uncertainties and important factors that may cause actual events or results to differ materially from those expressed or implied by any forward-looking statements including, without limitation, risks associated with: the timing and anticipated results of our current and future preclinical studies and clinical trials, supply chain, strategy and future operations; the delay of any current and future preclinical studies or clinical trials or the development of our product candidates; the risk that the results of prior preclinical studies and clinical trials may not be predictive of future results in connection with current or future preclinical studies and clinical trials, including those for abdakibart, the risk that cross-trial comparisons may not be reliable as no head-to-head trials have been conducted comparing abdakibart to lutikizumab, sonelokimab, povorcitinib, bimekizumab, secukinumab and adalimumab, and Phase 3 clinical data for abdakibart may not be directly comparable to clinical data of lutikizumab, sonelokimab, povorcitinib, bimekizumab, secukinumab and adalimumab due to differences in molecule composition, trial protocols, dosing regimens, and patient populations and characteristics; the timing and outcome of any interactions with regulatory authorities; obtaining, maintaining and protecting our intellectual property; the availability of funding sufficient for our operating expenses and capital expenditure requirements, reliance on key personnel; regulatory risks; general economic and market risks and uncertainties, including those caused by the war in Ukraine and the Middle East; and those other risks detailed in our filings with the Securities and Exchange Commission, available at www.sec.gov. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements, and you should not place undue reliance on our forward-looking statements. In addition, any forward-looking statements represent our view only as of today and should not be relied upon as representing its views as of any subsequent date. You should not rely upon forward-looking statements as predictions of future events and actual results or events could differ materially from the plans, intentions and expectations disclosed herein. Except as required by applicable law, we expressly disclaim any obligations or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in our expectations with respect thereto or any change in events, conditions or circumstances on which any statement is based. Certain information contained in this presentation and statements made orally during this presentation relate to or is based on studies, publications, surveys and other data obtained from third-party sources and our own internal estimates and research. This presentation contains trademarks, trade names and service marks of other companies, which are the property of their respective owners. 2

Building a New Standard in HS 3 HS, hidradenitis suppurativa; IL, interleukin; HiSCR, Hidradenitis Suppurativa Clinical Response. Note: The combined HiSCR50 abdakibart versus placebo analysis was performed post-hoc; Limitations exist in cross-trial comparisons across different phases of development. Compelling Efficacy • 42.5% (p=0.004) combined HiSCR75 and 61.7% (p=0.0009) combined HiSCR50, the highest absolute response rates observed in a study of this size or larger Consistent Response • All secondary endpoints were statistically significant or numerically favorable • Response rates similar across doses and regardless of prior biologic exposure Favorable Safety • Abdakibart was well-tolerated. No adverse events related to neutropenia, serious or opportunistic infections Simple Monthly Dosing • Potential for differentiated and patient friendly monthly dosing regimen starting at treatment initiation Abdakibart (AVTX-009) highly potent, specific inhibitor of IL-1β Avalo plans to initiate a Phase 3 registrational program in HS NEXT STEPS
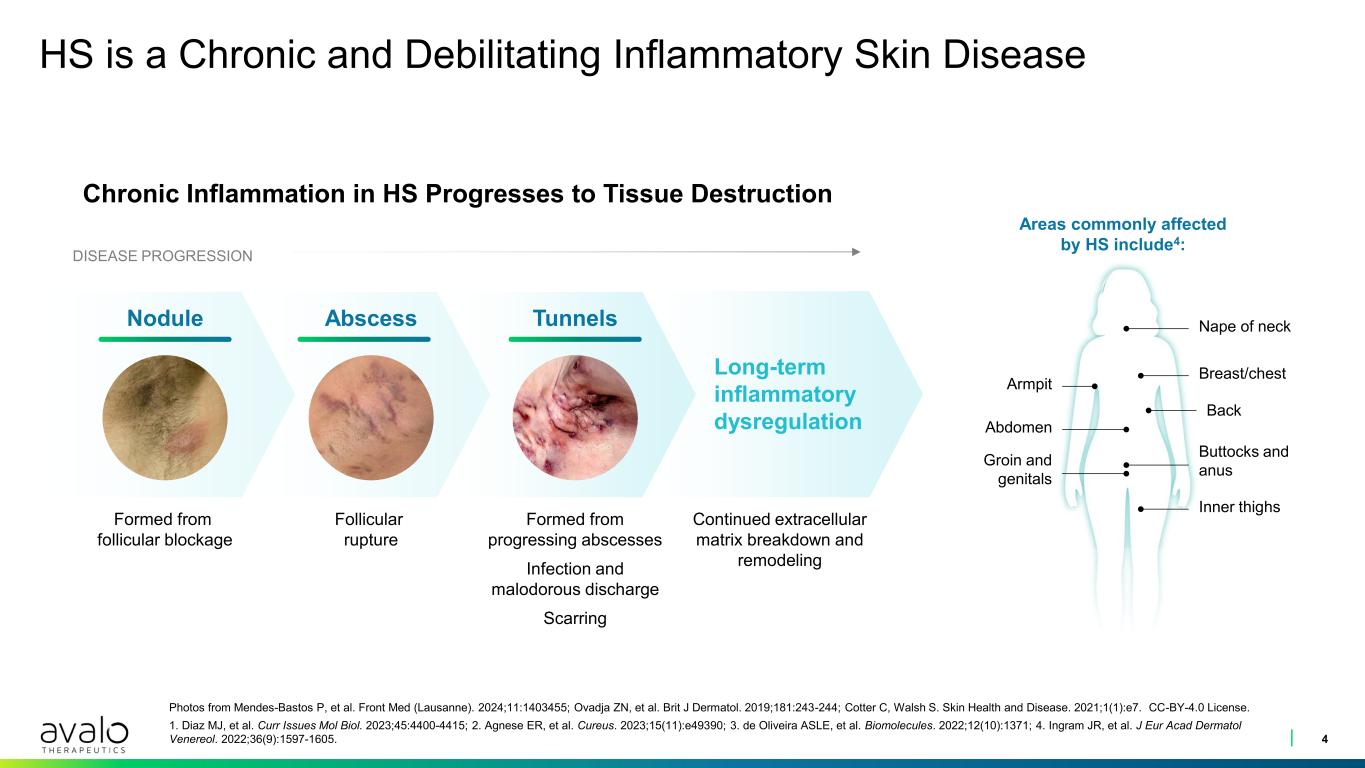
Nape of neck Breast/chest Buttocks and anus Inner thighs Groin and genitals Abdomen Armpit HS is a Chronic and Debilitating Inflammatory Skin Disease 4 Photos from Mendes-Bastos P, et al. Front Med (Lausanne). 2024;11:1403455; Ovadja ZN, et al. Brit J Dermatol. 2019;181:243-244; Cotter C, Walsh S. Skin Health and Disease. 2021;1(1):e7. CC-BY-4.0 License. 1. Diaz MJ, et al. Curr Issues Mol Biol. 2023;45:4400-4415; 2. Agnese ER, et al. Cureus. 2023;15(11):e49390; 3. de Oliveira ASLE, et al. Biomolecules. 2022;12(10):1371; 4. Ingram JR, et al. J Eur Acad Dermatol Venereol. 2022;36(9):1597-1605. Long-term inflammatory dysregulation Formed from progressing abscesses Infection and malodorous discharge Scarring Follicular rupture Formed from follicular blockage Areas commonly affected by HS include4: DISEASE PROGRESSION TunnelsAbscessNodule Continued extracellular matrix breakdown and remodeling Back Chronic Inflammation in HS Progresses to Tissue Destruction
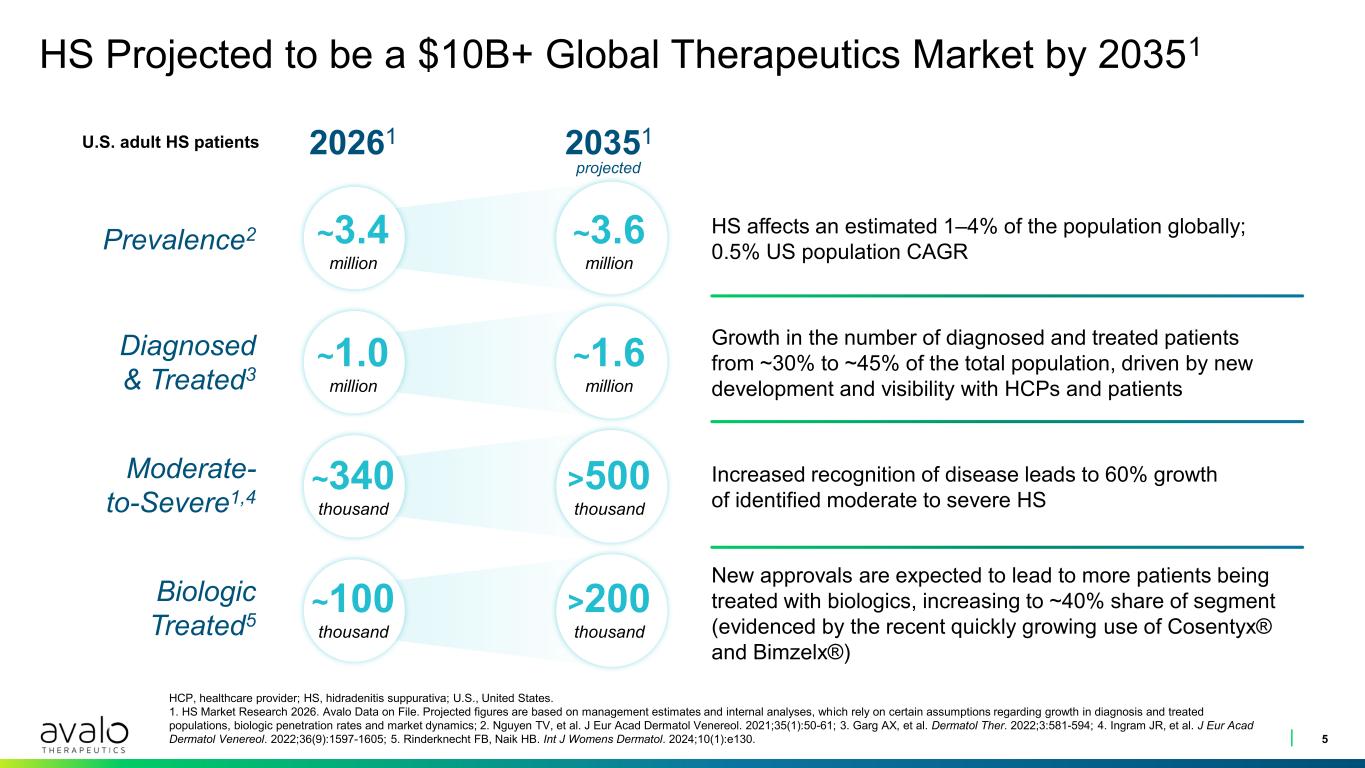
HS Projected to be a $10B+ Global Therapeutics Market by 20351 5 HCP, healthcare provider; HS, hidradenitis suppurativa; U.S., United States. 1. HS Market Research 2026. Avalo Data on File. Projected figures are based on management estimates and internal analyses, which rely on certain assumptions regarding growth in diagnosis and treated populations, biologic penetration rates and market dynamics; 2. Nguyen TV, et al. J Eur Acad Dermatol Venereol. 2021;35(1):50-61; 3. Garg AX, et al. Dermatol Ther. 2022;3:581-594; 4. Ingram JR, et al. J Eur Acad Dermatol Venereol. 2022;36(9):1597-1605; 5. Rinderknecht FB, Naik HB. Int J Womens Dermatol. 2024;10(1):e130. U.S. adult HS patients 20261 20351 Prevalence2 ~3.4 million ~3.6 million Diagnosed & Treated3 ~1.0 million ~1.6 million Moderate- to-Severe1,4 ~340 thousand >500 thousand Biologic Treated5 ~100 thousand >200 thousand HS affects an estimated 1–4% of the population globally; 0.5% US population CAGR Growth in the number of diagnosed and treated patients from ~30% to ~45% of the total population, driven by new development and visibility with HCPs and patients New approvals are expected to lead to more patients being treated with biologics, increasing to ~40% share of segment (evidenced by the recent quickly growing use of Cosentyx® and Bimzelx®) Increased recognition of disease leads to 60% growth of identified moderate to severe HS projected
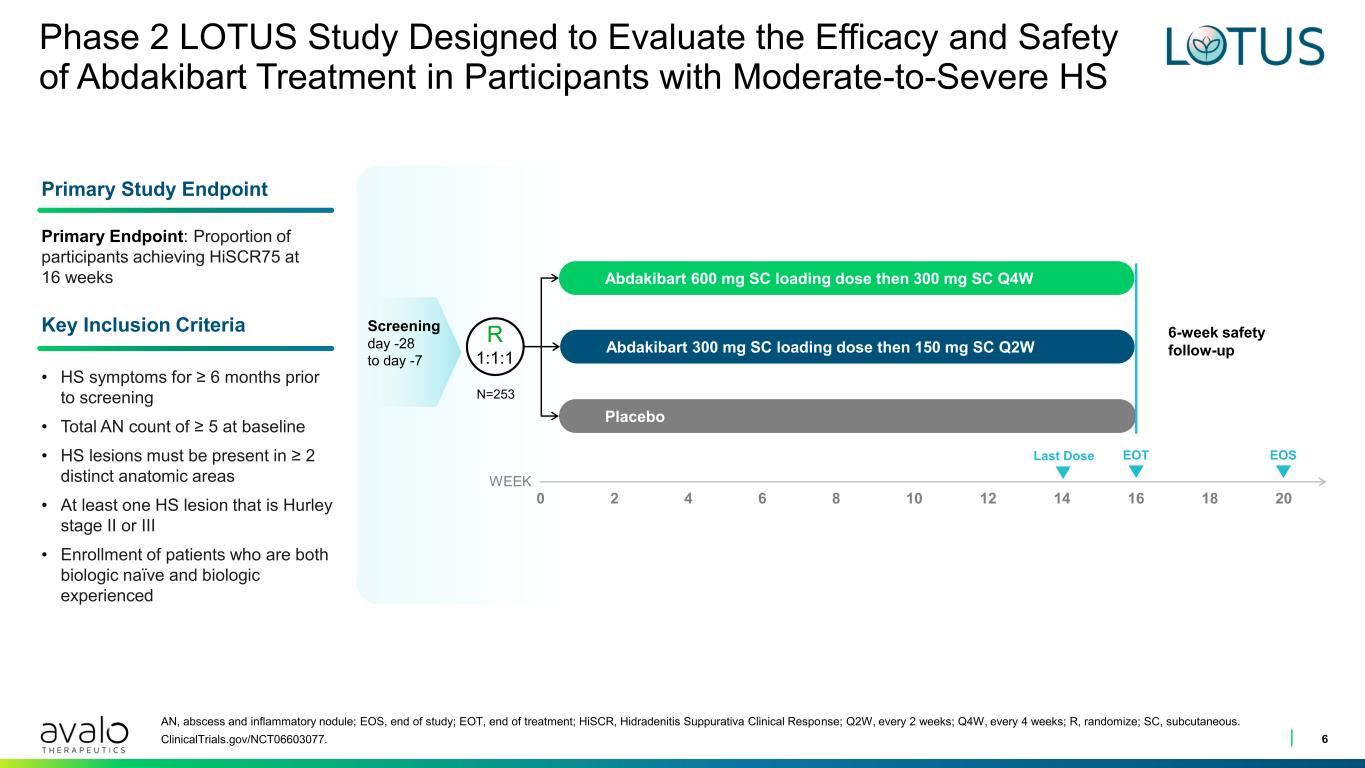
Phase 2 LOTUS Study Designed to Evaluate the Efficacy and Safety of Abdakibart Treatment in Participants with Moderate-to-Severe HS AN, abscess and inflammatory nodule; EOS, end of study; EOT, end of treatment; HiSCR, Hidradenitis Suppurativa Clinical Response; Q2W, every 2 weeks; Q4W, every 4 weeks; R, randomize; SC, subcutaneous. ClinicalTrials.gov/NCT06603077. • HS symptoms for ≥ 6 months prior to screening • Total AN count of ≥ 5 at baseline • HS lesions must be present in ≥ 2 distinct anatomic areas • At least one HS lesion that is Hurley stage II or III • Enrollment of patients who are both biologic naïve and biologic experienced Primary Endpoint: Proportion of participants achieving HiSCR75 at 16 weeks Abdakibart 600 mg SC loading dose then 300 mg SC Q4W Abdakibart 300 mg SC loading dose then 150 mg SC Q2W Placebo 6-week safety follow-up Screening day -28 to day -7 EOT WEEK Primary Study Endpoint Key Inclusion Criteria R 1:1:1 N=253 0 2 4 6 8 10 12 14 16 18 20 EOSLast Dose 6
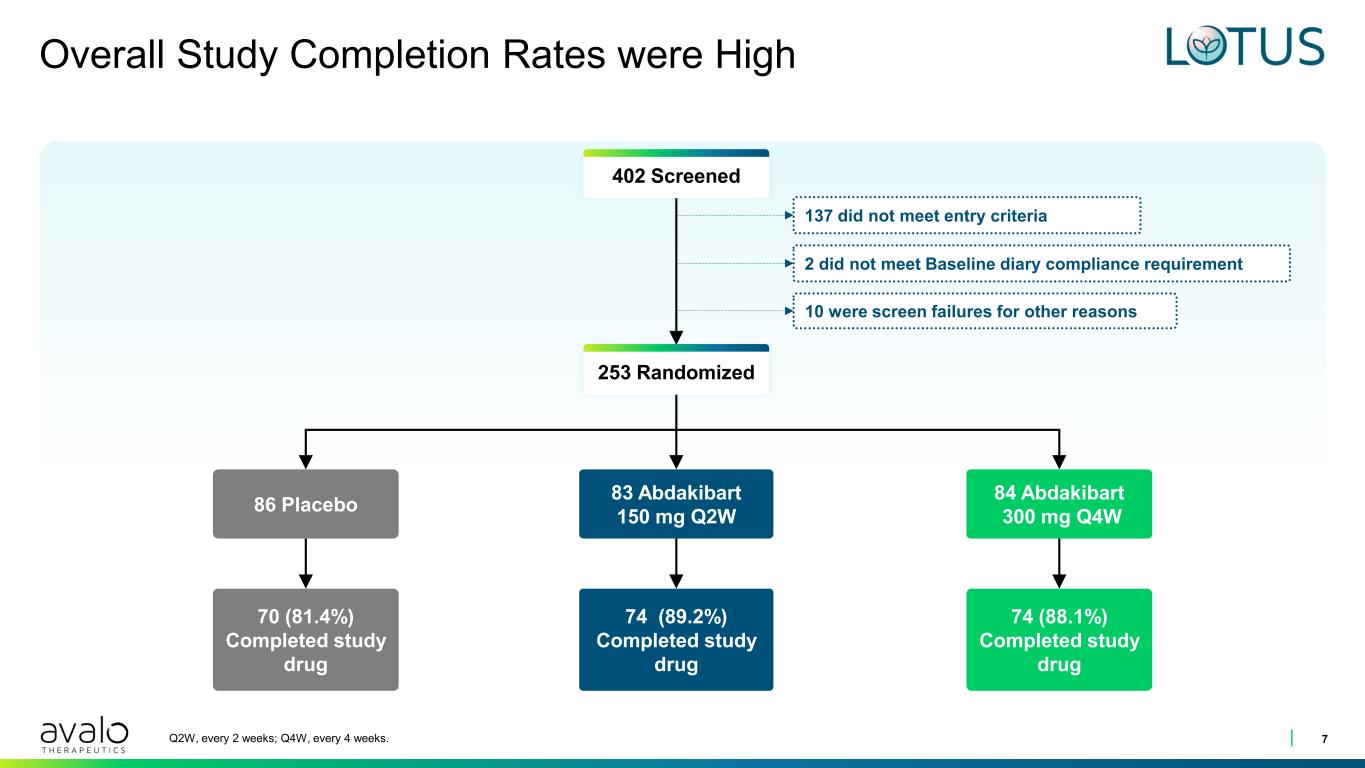
Overall Study Completion Rates were High 7Q2W, every 2 weeks; Q4W, every 4 weeks. 137 did not meet entry criteria 2 did not meet Baseline diary compliance requirement 10 were screen failures for other reasons 86 Placebo 83 Abdakibart 150 mg Q2W 84 Abdakibart 300 mg Q4W 70 (81.4%) Completed study drug 74 (89.2%) Completed study drug 74 (88.1%) Completed study drug 253 Randomized 402 Screened
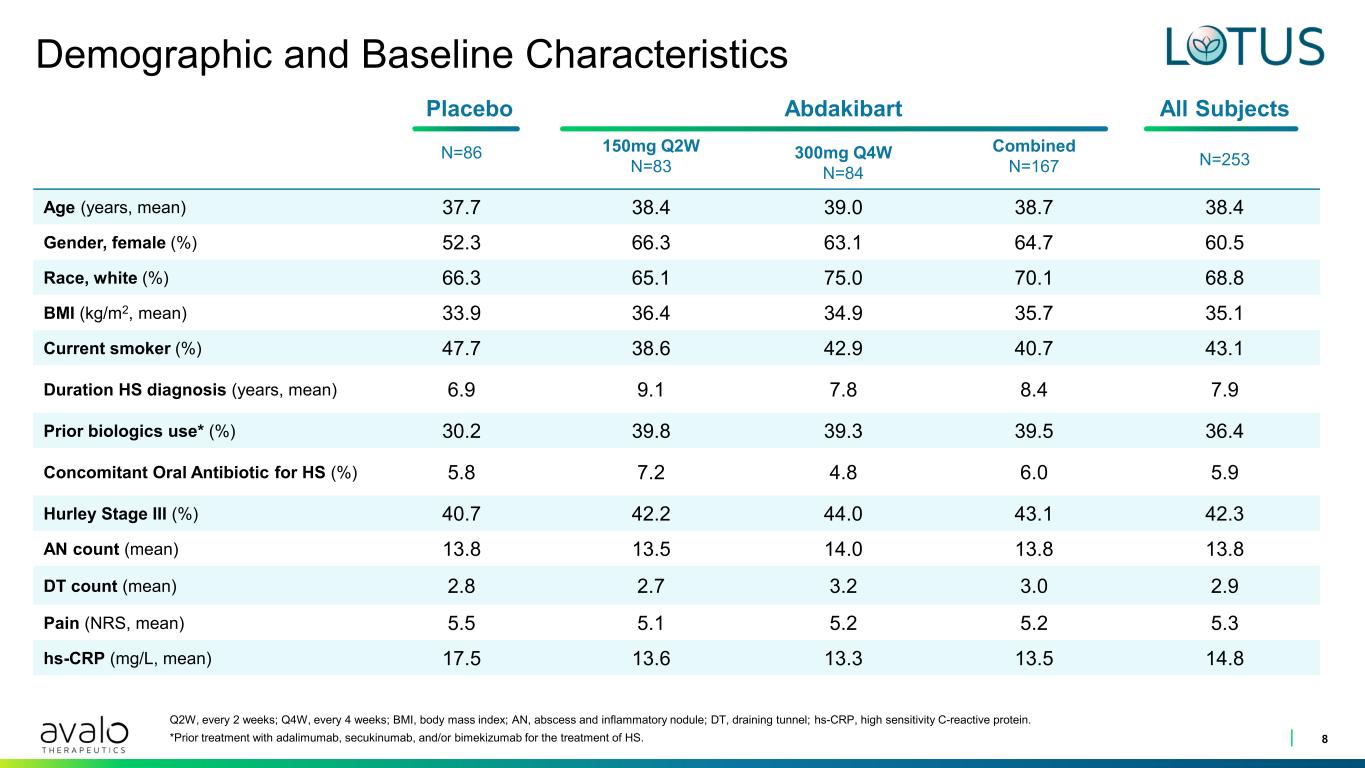
Placebo N=86 150mg Q2W N=83 Abdakibart 300mg Q4W N=84 Combined N=167 All Subjects N=253 Age (years, mean) 37.7 38.4 39.0 38.7 38.4 Gender, female (%) 52.3 66.3 63.1 64.7 60.5 Race, white (%) 66.3 65.1 75.0 70.1 68.8 BMI (kg/m2, mean) 33.9 36.4 34.9 35.7 35.1 Current smoker (%) 47.7 38.6 42.9 40.7 43.1 Duration HS diagnosis (years, mean) 6.9 9.1 7.8 8.4 7.9 Prior biologics use* (%) 30.2 39.8 39.3 39.5 36.4 Concomitant Oral Antibiotic for HS (%) 5.8 7.2 4.8 6.0 5.9 Hurley Stage III (%) 40.7 42.2 44.0 43.1 42.3 AN count (mean) 13.8 13.5 14.0 13.8 13.8 DT count (mean) 2.8 2.7 3.2 3.0 2.9 Pain (NRS, mean) 5.5 5.1 5.2 5.2 5.3 hs-CRP (mg/L, mean) 17.5 13.6 13.3 13.5 14.8 Demographic and Baseline Characteristics 8 Q2W, every 2 weeks; Q4W, every 4 weeks; BMI, body mass index; AN, abscess and inflammatory nodule; DT, draining tunnel; hs-CRP, high sensitivity C-reactive protein. *Prior treatment with adalimumab, secukinumab, and/or bimekizumab for the treatment of HS.
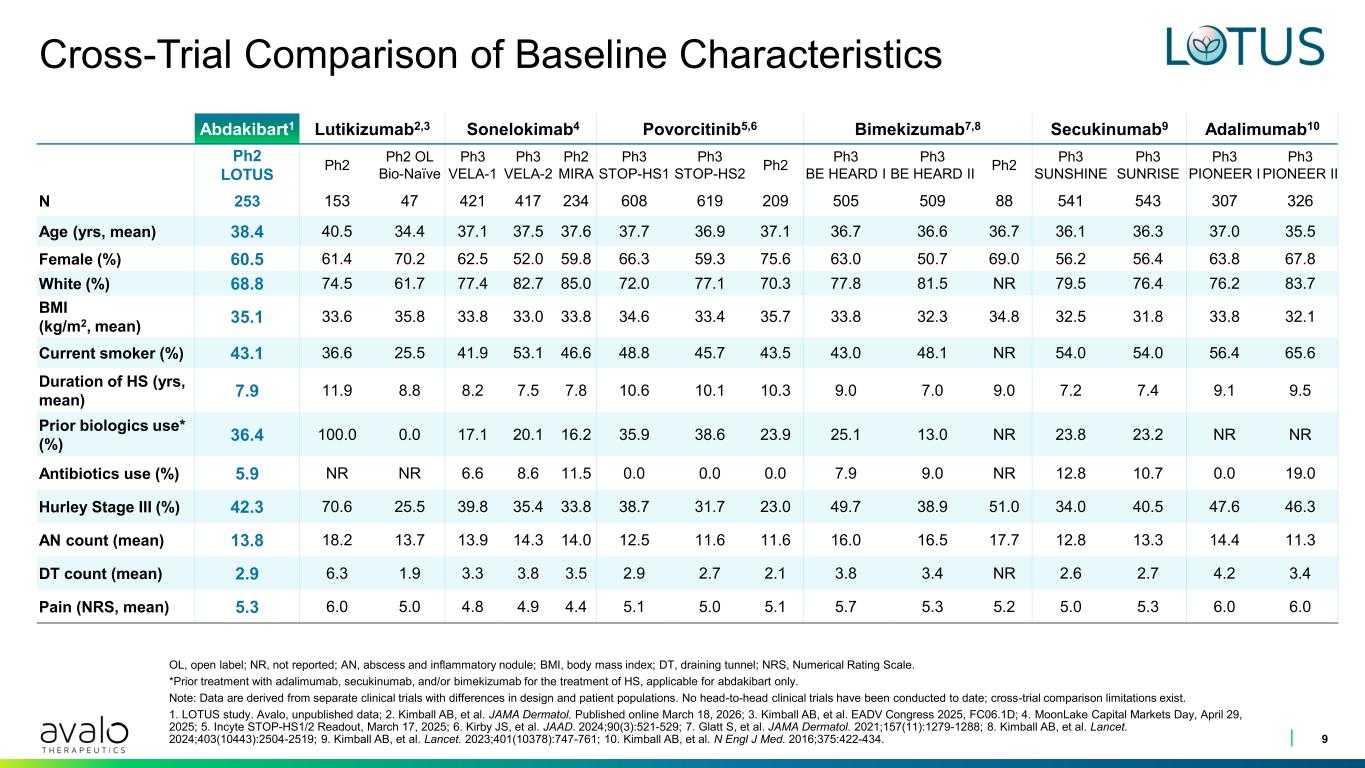
Abdakibart1 Lutikizumab2,3 Sonelokimab4 Povorcitinib5,6 Bimekizumab7,8 Secukinumab9 Adalimumab10 Ph2 LOTUS Ph2 Ph2 OL Bio-Naïve Ph3 VELA-1 Ph3 VELA-2 Ph2 MIRA Ph3 STOP-HS1 Ph3 STOP-HS2 Ph2 Ph3 BE HEARD I Ph3 BE HEARD II Ph2 Ph3 SUNSHINE Ph3 SUNRISE Ph3 PIONEER I Ph3 PIONEER II N 253 153 47 421 417 234 608 619 209 505 509 88 541 543 307 326 Age (yrs, mean) 38.4 40.5 34.4 37.1 37.5 37.6 37.7 36.9 37.1 36.7 36.6 36.7 36.1 36.3 37.0 35.5 Female (%) 60.5 61.4 70.2 62.5 52.0 59.8 66.3 59.3 75.6 63.0 50.7 69.0 56.2 56.4 63.8 67.8 White (%) 68.8 74.5 61.7 77.4 82.7 85.0 72.0 77.1 70.3 77.8 81.5 NR 79.5 76.4 76.2 83.7 BMI (kg/m2, mean) 35.1 33.6 35.8 33.8 33.0 33.8 34.6 33.4 35.7 33.8 32.3 34.8 32.5 31.8 33.8 32.1 Current smoker (%) 43.1 36.6 25.5 41.9 53.1 46.6 48.8 45.7 43.5 43.0 48.1 NR 54.0 54.0 56.4 65.6 Duration of HS (yrs, mean) 7.9 11.9 8.8 8.2 7.5 7.8 10.6 10.1 10.3 9.0 7.0 9.0 7.2 7.4 9.1 9.5 Prior biologics use* (%) 36.4 100.0 0.0 17.1 20.1 16.2 35.9 38.6 23.9 25.1 13.0 NR 23.8 23.2 NR NR Antibiotics use (%) 5.9 NR NR 6.6 8.6 11.5 0.0 0.0 0.0 7.9 9.0 NR 12.8 10.7 0.0 19.0 Hurley Stage III (%) 42.3 70.6 25.5 39.8 35.4 33.8 38.7 31.7 23.0 49.7 38.9 51.0 34.0 40.5 47.6 46.3 AN count (mean) 13.8 18.2 13.7 13.9 14.3 14.0 12.5 11.6 11.6 16.0 16.5 17.7 12.8 13.3 14.4 11.3 DT count (mean) 2.9 6.3 1.9 3.3 3.8 3.5 2.9 2.7 2.1 3.8 3.4 NR 2.6 2.7 4.2 3.4 Pain (NRS, mean) 5.3 6.0 5.0 4.8 4.9 4.4 5.1 5.0 5.1 5.7 5.3 5.2 5.0 5.3 6.0 6.0 Cross-Trial Comparison of Baseline Characteristics 9 OL, open label; NR, not reported; AN, abscess and inflammatory nodule; BMI, body mass index; DT, draining tunnel; NRS, Numerical Rating Scale. *Prior treatment with adalimumab, secukinumab, and/or bimekizumab for the treatment of HS, applicable for abdakibart only. Note: Data are derived from separate clinical trials with differences in design and patient populations. No head-to-head clinical trials have been conducted to date; cross-trial comparison limitations exist. 1. LOTUS study. Avalo, unpublished data; 2. Kimball AB, et al. JAMA Dermatol. Published online March 18, 2026; 3. Kimball AB, et al. EADV Congress 2025, FC06.1D; 4. MoonLake Capital Markets Day, April 29, 2025; 5. Incyte STOP-HS1/2 Readout, March 17, 2025; 6. Kirby JS, et al. JAAD. 2024;90(3):521-529; 7. Glatt S, et al. JAMA Dermatol. 2021;157(11):1279-1288; 8. Kimball AB, et al. Lancet. 2024;403(10443):2504-2519; 9. Kimball AB, et al. Lancet. 2023;401(10378):747-761; 10. Kimball AB, et al. N Engl J Med. 2016;375:422-434.
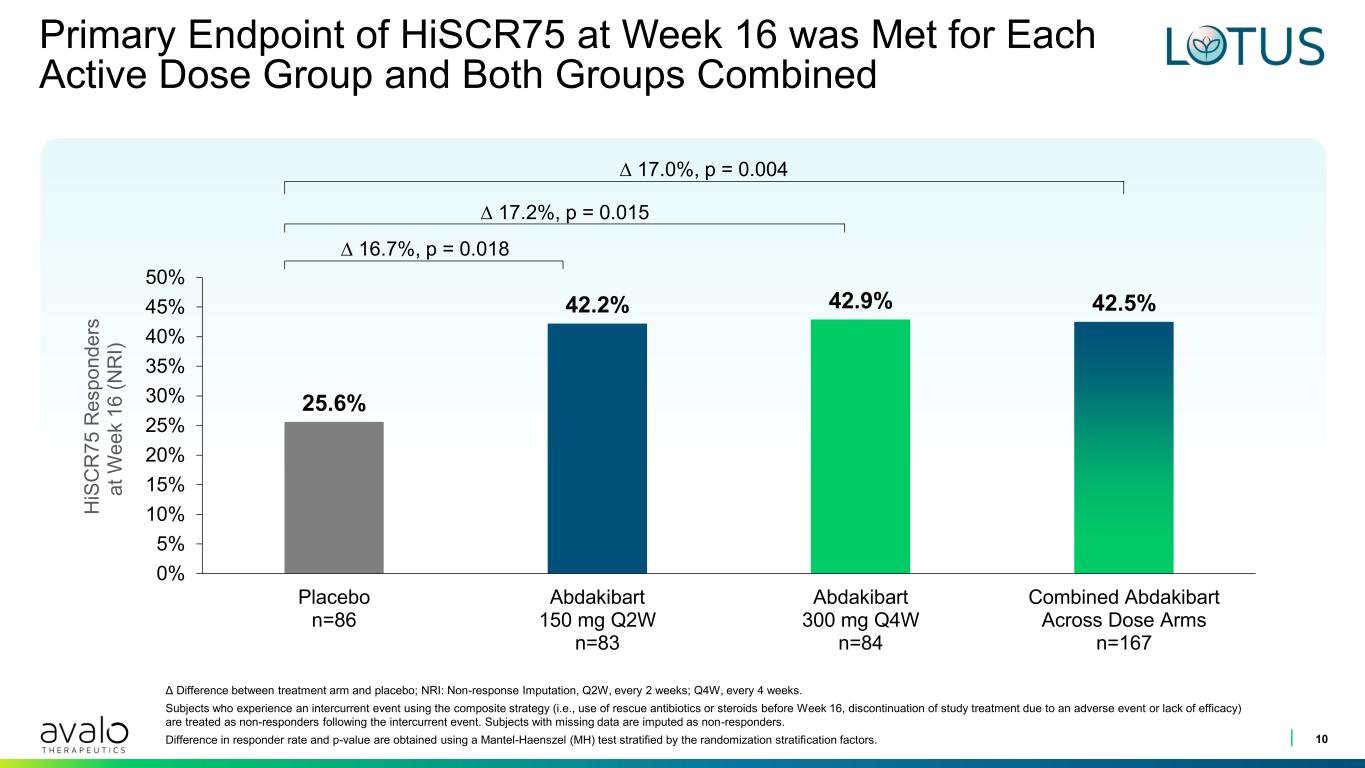
Primary Endpoint of HiSCR75 at Week 16 was Met for Each Active Dose Group and Both Groups Combined 10 25.6% 42.2% 42.9% 42.5% 0% 5% 10% 15% 20% 25% 30% 35% 40% 45% 50% Placebo n=86 Abdakibart 150 mg Q2W n=83 Abdakibart 300 mg Q4W n=84 Combined Abdakibart Across Dose Arms n=167 H iS C R 7 5 R e s p o n d e rs a t W e e k 1 6 ( N R I) ∆ 17.0%, p = 0.004 ∆ 17.2%, p = 0.015 ∆ 16.7%, p = 0.018 Δ Difference between treatment arm and placebo; NRI: Non-response Imputation, Q2W, every 2 weeks; Q4W, every 4 weeks. Subjects who experience an intercurrent event using the composite strategy (i.e., use of rescue antibiotics or steroids before Week 16, discontinuation of study treatment due to an adverse event or lack of efficacy) are treated as non-responders following the intercurrent event. Subjects with missing data are imputed as non-responders. Difference in responder rate and p-value are obtained using a Mantel-Haenszel (MH) test stratified by the randomization stratification factors.
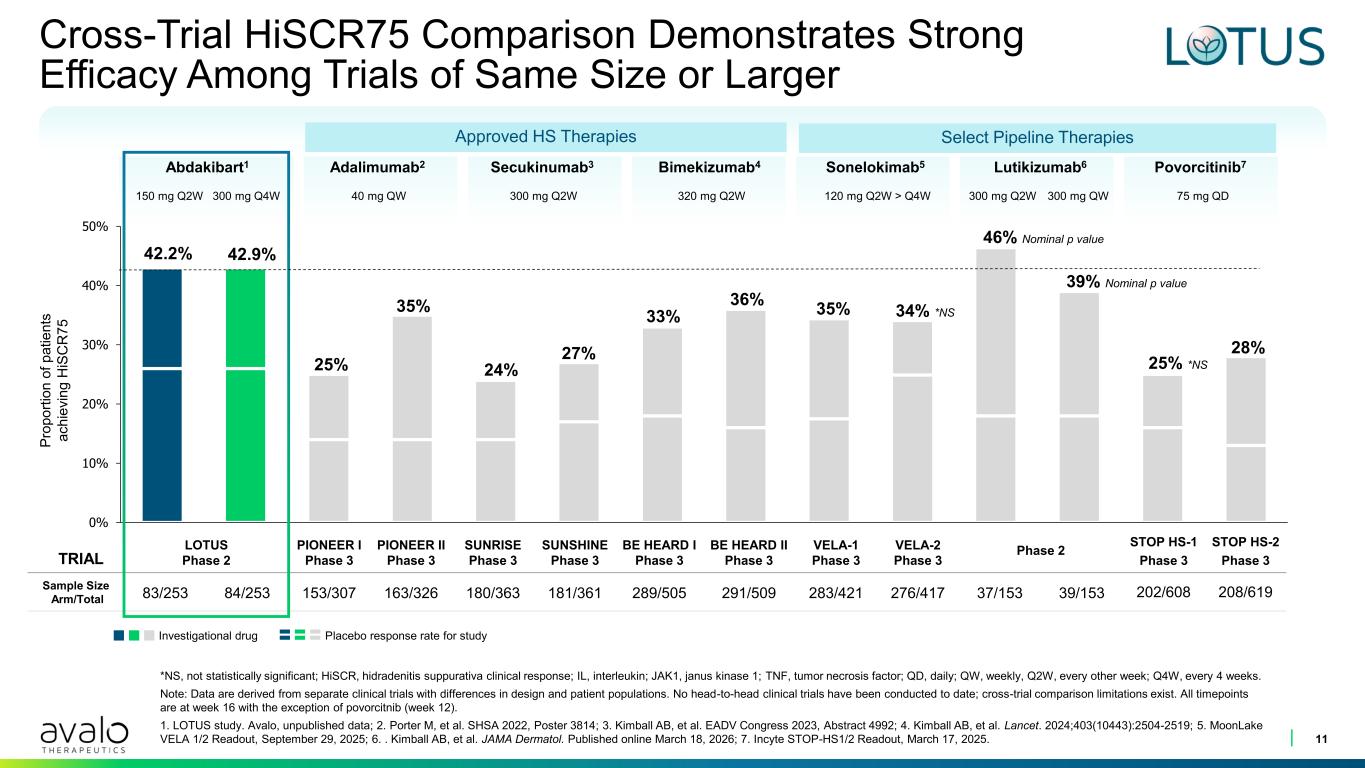
0% 10% 20% 30% 40% 50% TRIAL LOTUS Phase 2 PIONEER I Phase 3 PIONEER II Phase 3 SUNRISE Phase 3 SUNSHINE Phase 3 BE HEARD I Phase 3 BE HEARD II Phase 3 VELA-1 Phase 3 VELA-2 Phase 3 Phase 2 STOP HS-1 Phase 3 STOP HS-2 Phase 3 Sample Size Arm/Total 83/253 84/253 153/307 163/326 180/363 181/361 289/505 291/509 283/421 276/417 37/153 39/153 202/608 208/619 Cross-Trial HiSCR75 Comparison Demonstrates Strong Efficacy Among Trials of Same Size or Larger 11 *NS, not statistically significant; HiSCR, hidradenitis suppurativa clinical response; IL, interleukin; JAK1, janus kinase 1; TNF, tumor necrosis factor; QD, daily; QW, weekly, Q2W, every other week; Q4W, every 4 weeks. Note: Data are derived from separate clinical trials with differences in design and patient populations. No head-to-head clinical trials have been conducted to date; cross-trial comparison limitations exist. All timepoints are at week 16 with the exception of povorcitnib (week 12). 1. LOTUS study. Avalo, unpublished data; 2. Porter M, et al. SHSA 2022, Poster 3814; 3. Kimball AB, et al. EADV Congress 2023, Abstract 4992; 4. Kimball AB, et al. Lancet. 2024;403(10443):2504-2519; 5. MoonLake VELA 1/2 Readout, September 29, 2025; 6. . Kimball AB, et al. JAMA Dermatol. Published online March 18, 2026; 7. Incyte STOP-HS1/2 Readout, March 17, 2025. P ro p o rt io n o f p a ti e n ts a c h ie v in g H iS C R 7 5 Investigational drug Placebo response rate for study Sonelokimab5Bimekizumab4 Povorcitinib7Abdakibart1 Lutikizumab6Secukinumab3Adalimumab2 Approved HS Therapies 42.2% 42.9% 25% 35% 24% 27% 33% 36% 35% 34% *NS 46% Nominal p value 39% Nominal p value 25% *NS 28% Select Pipeline Therapies 120 mg Q2W > Q4W320 mg Q2W 75 mg QD300 mg Q2W300 mg Q4W150 mg Q2W 300 mg Q2W 300 mg QW40 mg QW
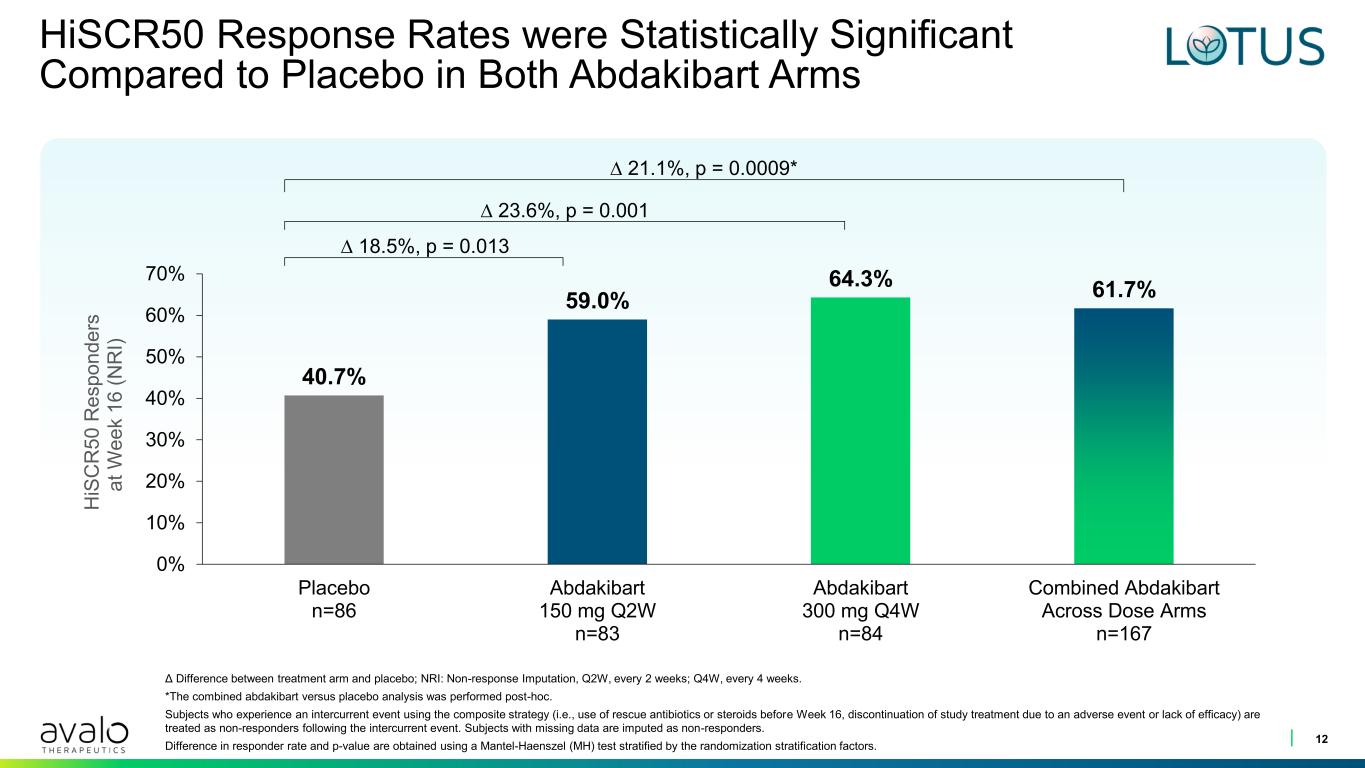
HiSCR50 Response Rates were Statistically Significant Compared to Placebo in Both Abdakibart Arms 12 40.7% 59.0% 64.3% 61.7% 0% 10% 20% 30% 40% 50% 60% 70% Placebo n=86 Abdakibart 150 mg Q2W n=83 Abdakibart 300 mg Q4W n=84 Combined Abdakibart Across Dose Arms n=167 H iS C R 5 0 R e s p o n d e rs a t W e e k 1 6 ( N R I) ∆ 21.1%, p = 0.0009* ∆ 23.6%, p = 0.001 ∆ 18.5%, p = 0.013 Δ Difference between treatment arm and placebo; NRI: Non-response Imputation, Q2W, every 2 weeks; Q4W, every 4 weeks. *The combined abdakibart versus placebo analysis was performed post-hoc. Subjects who experience an intercurrent event using the composite strategy (i.e., use of rescue antibiotics or steroids before Week 16, discontinuation of study treatment due to an adverse event or lack of efficacy) are treated as non-responders following the intercurrent event. Subjects with missing data are imputed as non-responders. Difference in responder rate and p-value are obtained using a Mantel-Haenszel (MH) test stratified by the randomization stratification factors.
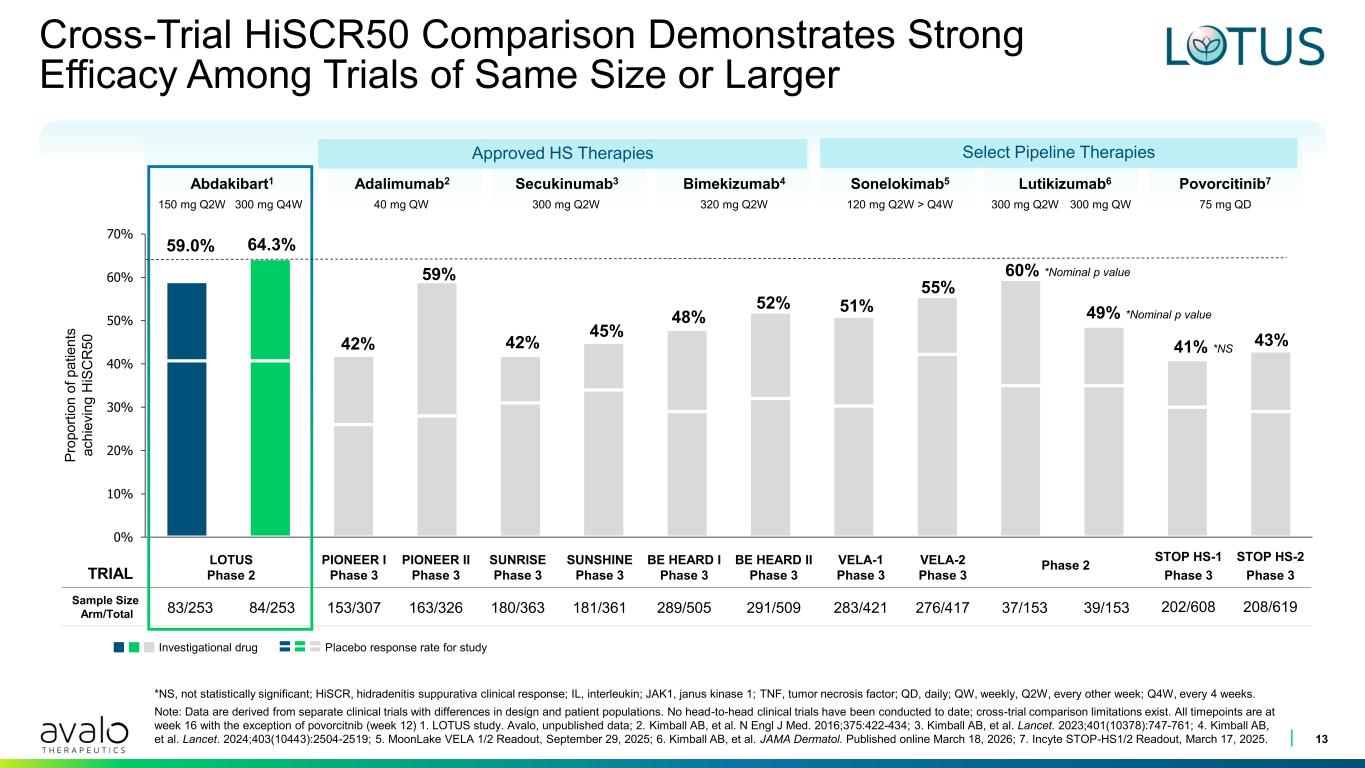
0% 10% 20% 30% 40% 50% 60% 70% TRIAL LOTUS Phase 2 PIONEER I Phase 3 PIONEER II Phase 3 SUNRISE Phase 3 SUNSHINE Phase 3 BE HEARD I Phase 3 BE HEARD II Phase 3 VELA-1 Phase 3 VELA-2 Phase 3 Phase 2 STOP HS-1 Phase 3 STOP HS-2 Phase 3 Sample Size Arm/Total 83/253 84/253 153/307 163/326 180/363 181/361 289/505 291/509 283/421 276/417 37/153 39/153 202/608 208/619 Cross-Trial HiSCR50 Comparison Demonstrates Strong Efficacy Among Trials of Same Size or Larger 13 *NS, not statistically significant; HiSCR, hidradenitis suppurativa clinical response; IL, interleukin; JAK1, janus kinase 1; TNF, tumor necrosis factor; QD, daily; QW, weekly, Q2W, every other week; Q4W, every 4 weeks. Note: Data are derived from separate clinical trials with differences in design and patient populations. No head-to-head clinical trials have been conducted to date; cross-trial comparison limitations exist. All timepoints are at week 16 with the exception of povorcitnib (week 12) 1. LOTUS study. Avalo, unpublished data; 2. Kimball AB, et al. N Engl J Med. 2016;375:422-434; 3. Kimball AB, et al. Lancet. 2023;401(10378):747-761; 4. Kimball AB, et al. Lancet. 2024;403(10443):2504-2519; 5. MoonLake VELA 1/2 Readout, September 29, 2025; 6. Kimball AB, et al. JAMA Dermatol. Published online March 18, 2026; 7. Incyte STOP-HS1/2 Readout, March 17, 2025. P ro p o rt io n o f p a ti e n ts a c h ie v in g H iS C R 5 0 Sonelokimab5 120 mg Q2W > Q4W Bimekizumab4 320 mg Q2W Povorcitinib7 75 mg QD300 mg Q2W Abdakibart1 300 mg Q4W150 mg Q2W Lutikizumab6 300 mg Q2W 300 mg QW Secukinumab3Adalimumab2 40 mg QW 42% 59% 42% 45% 48% 52% 51% 55% 60% *Nominal p value 49% *Nominal p value 41% *NS 43% 59.0% 64.3% Approved HS Therapies Select Pipeline Therapies Investigational drug Placebo response rate for study
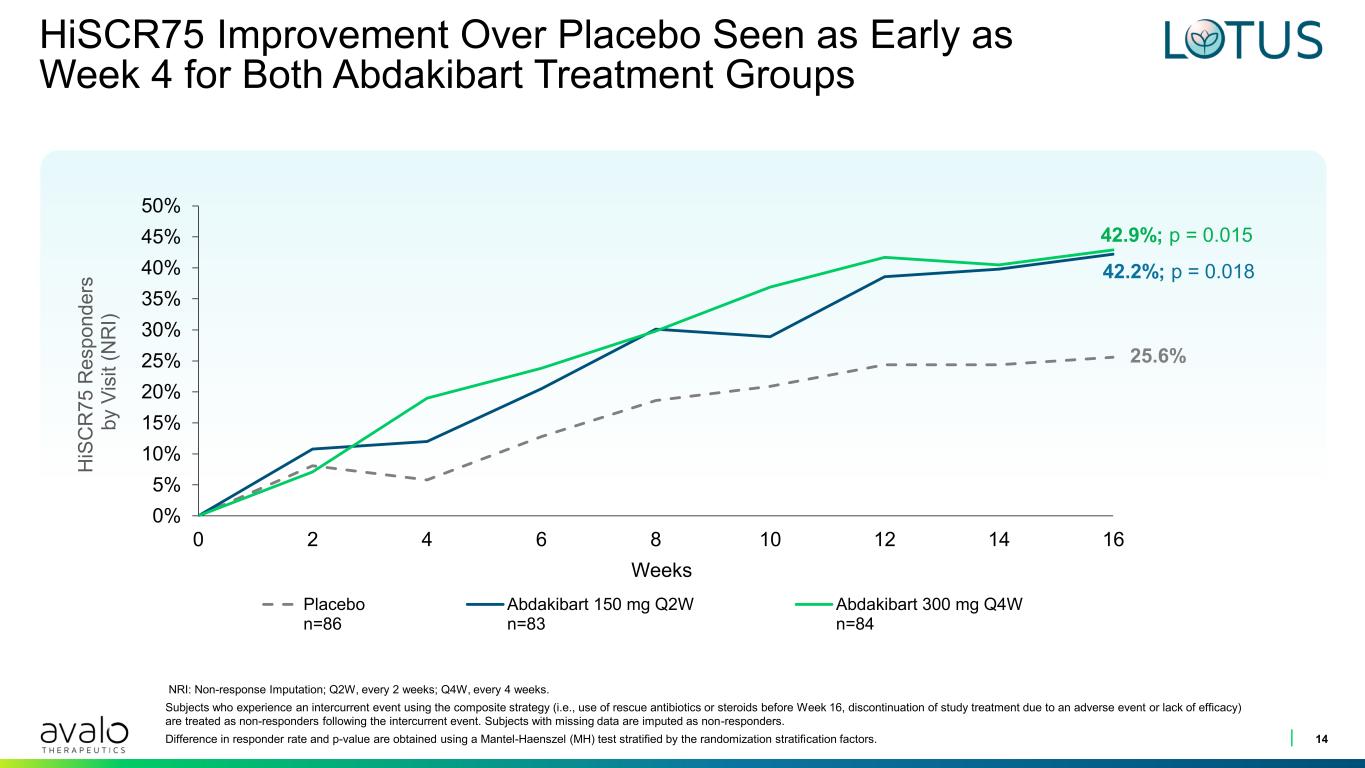
HiSCR75 Improvement Over Placebo Seen as Early as Week 4 for Both Abdakibart Treatment Groups 14 Weeks 0% 5% 10% 15% 20% 25% 30% 35% 40% 45% 50% 0 2 4 6 8 10 12 14 16 H iS C R 7 5 R e s p o n d e rs b y V is it ( N R I) Placebo n=86 Abdakibart 150 mg Q2W n=83 Abdakibart 300 mg Q4W n=84 42.9%; p = 0.015 42.2%; p = 0.018 25.6% NRI: Non-response Imputation; Q2W, every 2 weeks; Q4W, every 4 weeks. Subjects who experience an intercurrent event using the composite strategy (i.e., use of rescue antibiotics or steroids before Week 16, discontinuation of study treatment due to an adverse event or lack of efficacy) are treated as non-responders following the intercurrent event. Subjects with missing data are imputed as non-responders. Difference in responder rate and p-value are obtained using a Mantel-Haenszel (MH) test stratified by the randomization stratification factors.
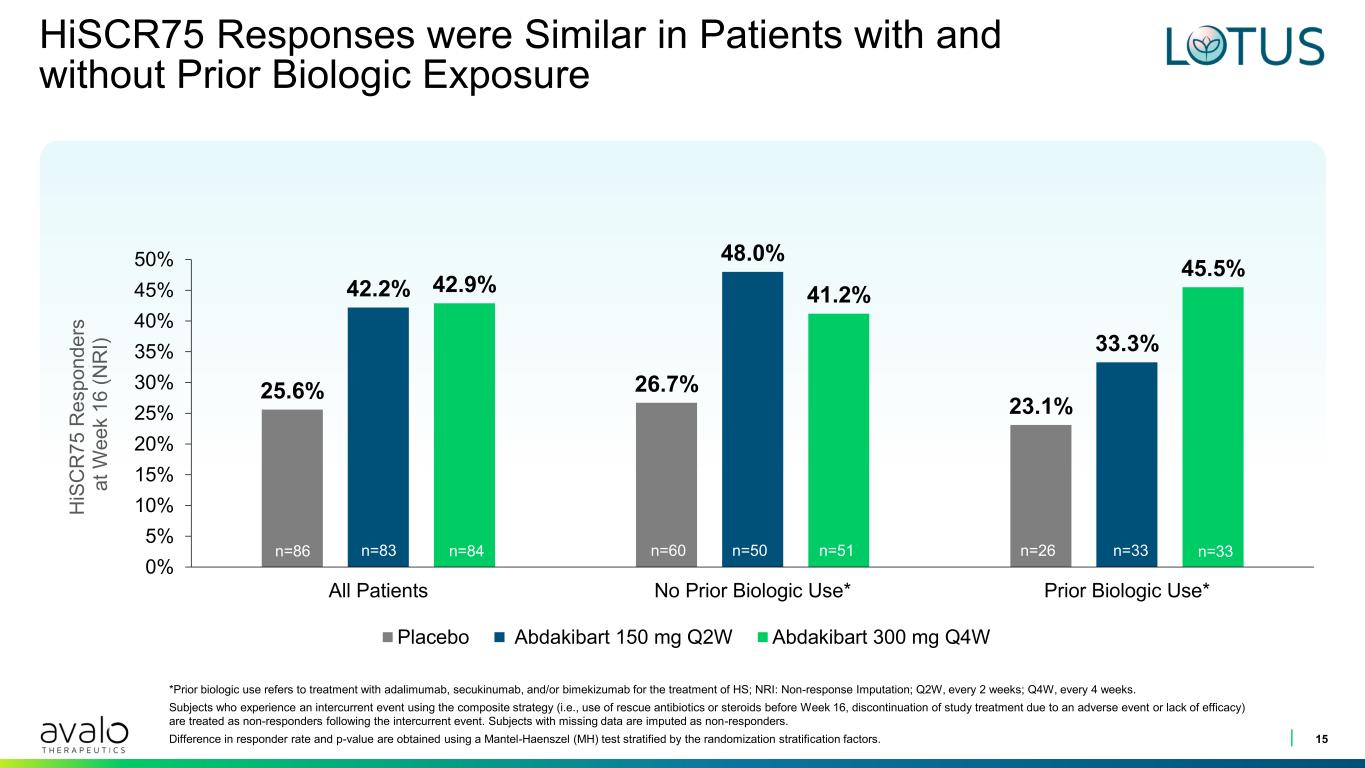
HiSCR75 Responses were Similar in Patients with and without Prior Biologic Exposure 15 25.6% 26.7% 23.1% 42.2% 48.0% 33.3% 42.9% 41.2% 45.5% 0% 5% 10% 15% 20% 25% 30% 35% 40% 45% 50% All Patients No Prior Biologic Use* Prior Biologic Use* H iS C R 7 5 R e s p o n d e rs a t W e e k 1 6 ( N R I) Placebo Abdakibart 150 mg Q2W Abdakibart 300 mg Q4W n=86 n=83 n=84 n=60 n=50 n=51 n=26 n=33 n=33 *Prior biologic use refers to treatment with adalimumab, secukinumab, and/or bimekizumab for the treatment of HS; NRI: Non-response Imputation; Q2W, every 2 weeks; Q4W, every 4 weeks. Subjects who experience an intercurrent event using the composite strategy (i.e., use of rescue antibiotics or steroids before Week 16, discontinuation of study treatment due to an adverse event or lack of efficacy) are treated as non-responders following the intercurrent event. Subjects with missing data are imputed as non-responders. Difference in responder rate and p-value are obtained using a Mantel-Haenszel (MH) test stratified by the randomization stratification factors.
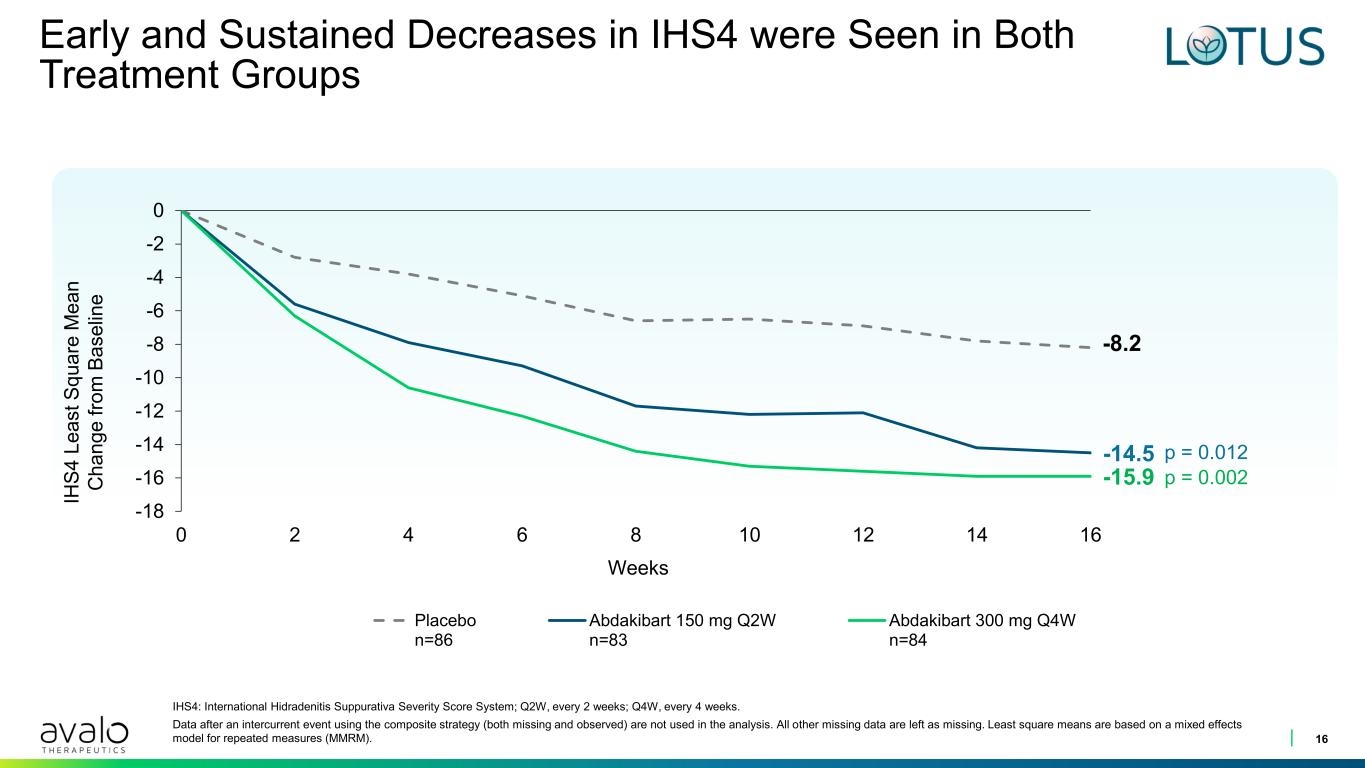
Early and Sustained Decreases in IHS4 were Seen in Both Treatment Groups 16 -8.2 -14.5 -15.9 -18 -16 -14 -12 -10 -8 -6 -4 -2 0 0 2 4 6 8 10 12 14 16 Placebo n=86 Abdakibart 150 mg Q2W n=83 Abdakibart 300 mg Q4W n=84 p = 0.002 p = 0.012 Weeks IHS4: International Hidradenitis Suppurativa Severity Score System; Q2W, every 2 weeks; Q4W, every 4 weeks. Data after an intercurrent event using the composite strategy (both missing and observed) are not used in the analysis. All other missing data are left as missing. Least square means are based on a mixed effects model for repeated measures (MMRM). IH S 4 L e a s t S q u a re M e a n C h a n g e f ro m B a s e lin e
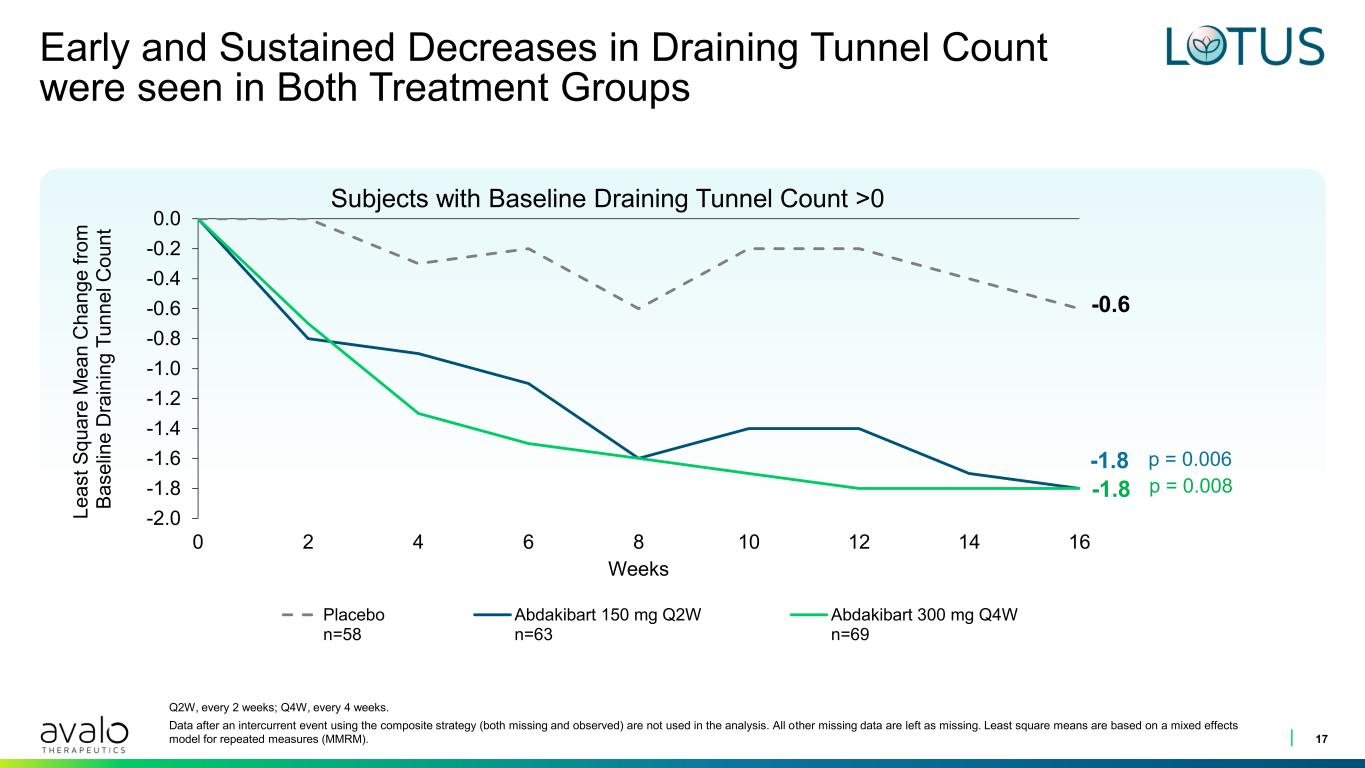
Early and Sustained Decreases in Draining Tunnel Count were seen in Both Treatment Groups 17 -0.6 -1.8 -2.0 -1.8 -1.6 -1.4 -1.2 -1.0 -0.8 -0.6 -0.4 -0.2 0.0 0 2 4 6 8 10 12 14 16 L e a s t S q u a re M e a n C h a n g e f ro m B a s e lin e D ra in in g T u n n e l C o u n t Weeks Placebo n=58 Abdakibart 150 mg Q2W n=63 Abdakibart 300 mg Q4W n=69 -1.8 p = 0.008 p = 0.006 Q2W, every 2 weeks; Q4W, every 4 weeks. Data after an intercurrent event using the composite strategy (both missing and observed) are not used in the analysis. All other missing data are left as missing. Least square means are based on a mixed effects model for repeated measures (MMRM). Subjects with Baseline Draining Tunnel Count >0
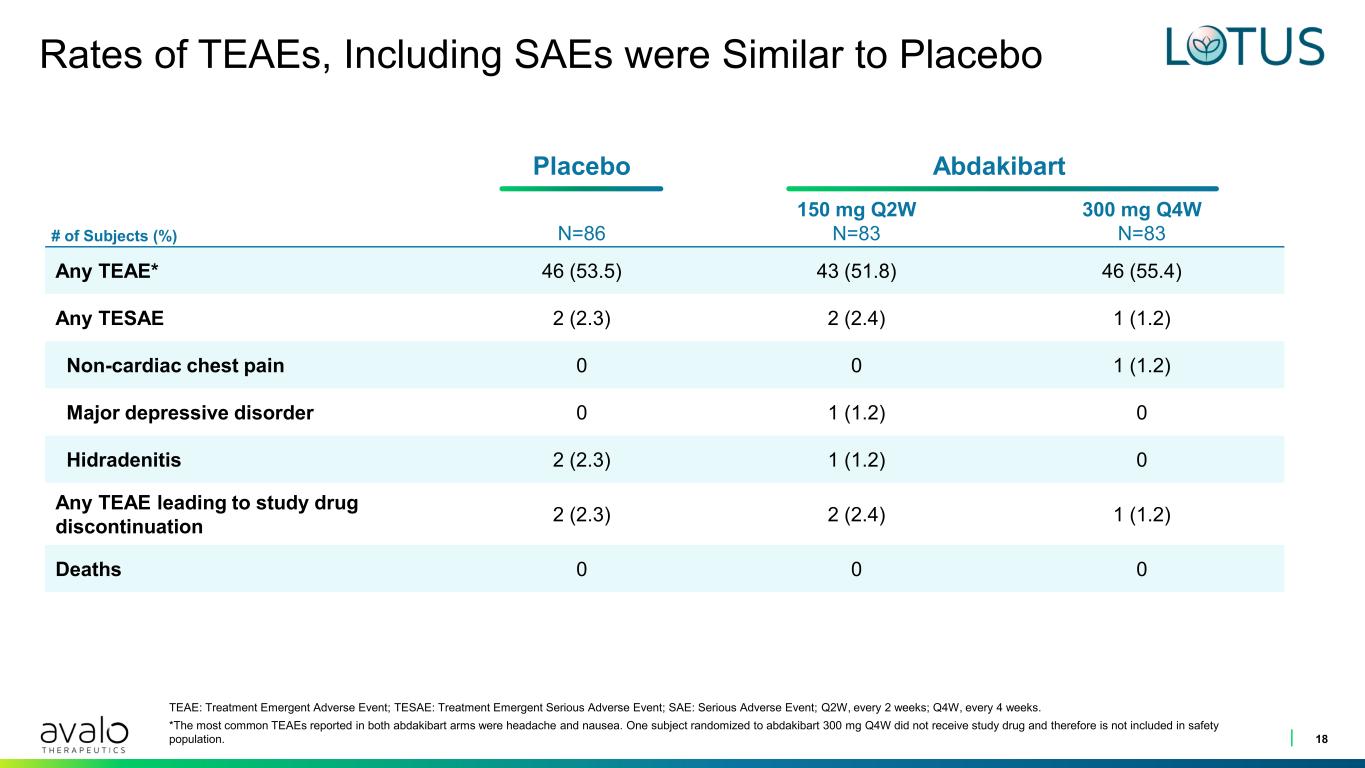
Placebo Abdakibart # of Subjects (%) N=86 150 mg Q2W N=83 300 mg Q4W N=83 Any TEAE* 46 (53.5) 43 (51.8) 46 (55.4) Any TESAE 2 (2.3) 2 (2.4) 1 (1.2) Non-cardiac chest pain 0 0 1 (1.2) Major depressive disorder 0 1 (1.2) 0 Hidradenitis 2 (2.3) 1 (1.2) 0 Any TEAE leading to study drug discontinuation 2 (2.3) 2 (2.4) 1 (1.2) Deaths 0 0 0 Rates of TEAEs, Including SAEs were Similar to Placebo 18 TEAE: Treatment Emergent Adverse Event; TESAE: Treatment Emergent Serious Adverse Event; SAE: Serious Adverse Event; Q2W, every 2 weeks; Q4W, every 4 weeks. *The most common TEAEs reported in both abdakibart arms were headache and nausea. One subject randomized to abdakibart 300 mg Q4W did not receive study drug and therefore is not included in safety population.
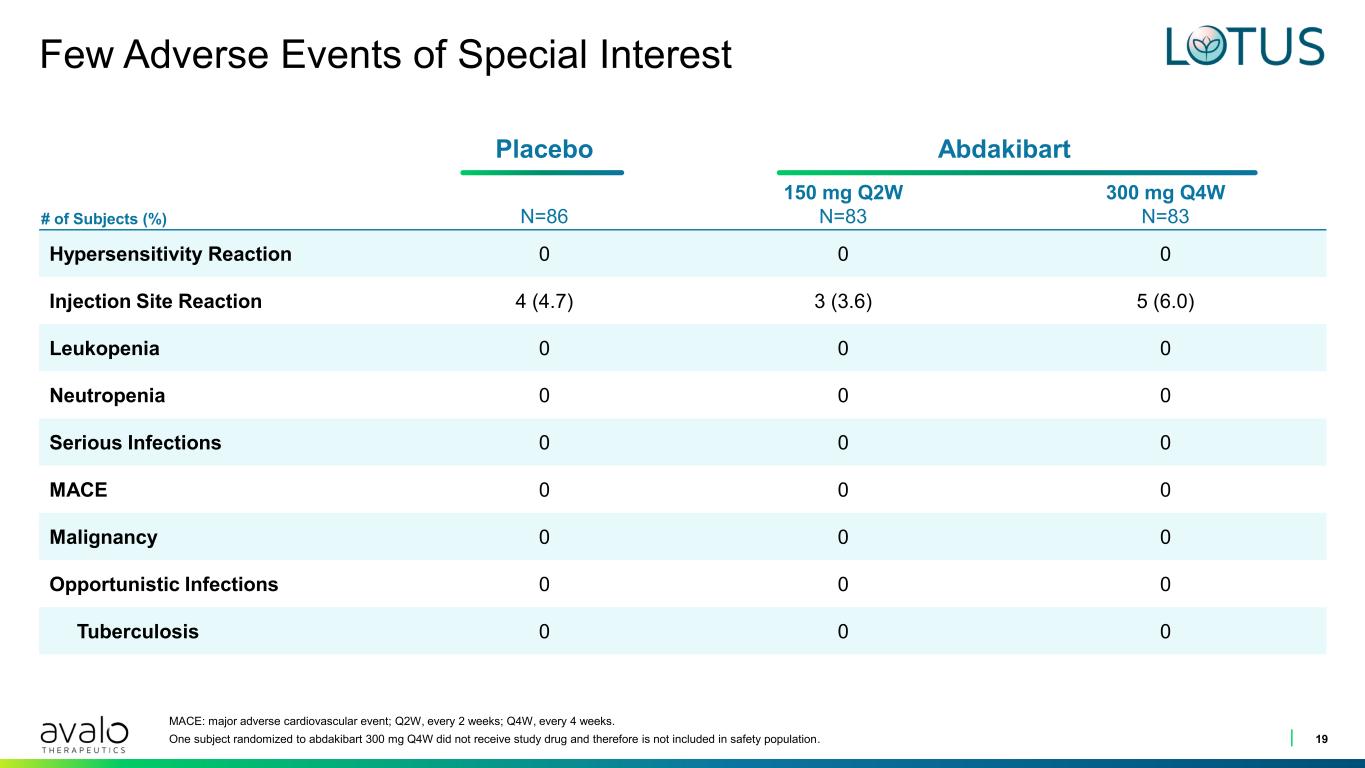
Placebo Abdakibart # of Subjects (%) N=86 150 mg Q2W N=83 300 mg Q4W N=83 Hypersensitivity Reaction 0 0 0 Injection Site Reaction 4 (4.7) 3 (3.6) 5 (6.0) Leukopenia 0 0 0 Neutropenia 0 0 0 Serious Infections 0 0 0 MACE 0 0 0 Malignancy 0 0 0 Opportunistic Infections 0 0 0 Tuberculosis 0 0 0 Few Adverse Events of Special Interest 19 MACE: major adverse cardiovascular event; Q2W, every 2 weeks; Q4W, every 4 weeks. One subject randomized to abdakibart 300 mg Q4W did not receive study drug and therefore is not included in safety population.

Building a New Standard in HS 20 HS, hidradenitis suppurativa; IL, interleukin; HiSCR, Hidradenitis Suppurativa Clinical Response. Note: The combined HiSCR50 abdakibart versus placebo analysis was performed post-hoc; Limitations exist in cross-trial comparisons across different phases of development. Compelling Efficacy • 42.5% (p=0.004) combined HiSCR75 and 61.7% (p=0.0009) combined HiSCR50, the highest absolute response rates observed in a study of this size or larger Consistent Response • All secondary endpoints were statistically significant or numerically favorable • Response rates similar across doses and regardless of prior biologic exposure Favorable Safety • Abdakibart was well-tolerated. No adverse events related to neutropenia, serious or opportunistic infections Simple Monthly Dosing • Potential for differentiated and patient friendly monthly dosing regimen starting at treatment initiation Abdakibart (AVTX-009) highly potent, specific inhibitor of IL-1β Avalo plans to initiate a Phase 3 registrational program in HS NEXT STEPS

Appendix 21
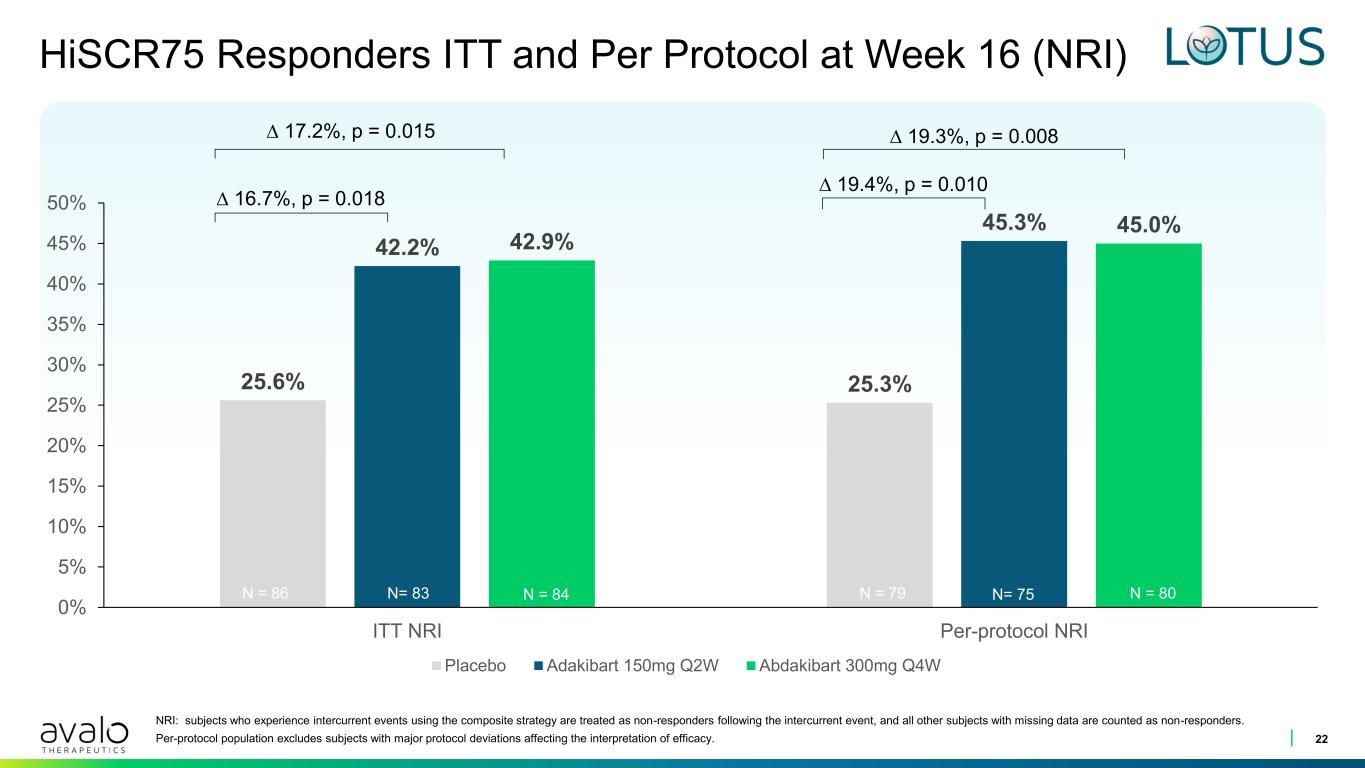
HiSCR75 Responders ITT and Per Protocol at Week 16 (NRI) 25.6% 25.3% 42.2% 45.3% 42.9% 45.0% 0% 5% 10% 15% 20% 25% 30% 35% 40% 45% 50% ITT NRI Per-protocol NRI Placebo Adakibart 150mg Q2W Abdakibart 300mg Q4W N= 75N = 79 N = 80 22 ∆ 19.4%, p = 0.010 ∆ 19.3%, p = 0.008∆ 17.2%, p = 0.015 ∆ 16.7%, p = 0.018 NRI: subjects who experience intercurrent events using the composite strategy are treated as non-responders following the intercurrent event, and all other subjects with missing data are counted as non-responders. Per-protocol population excludes subjects with major protocol deviations affecting the interpretation of efficacy. N= 83N = 86 N = 84
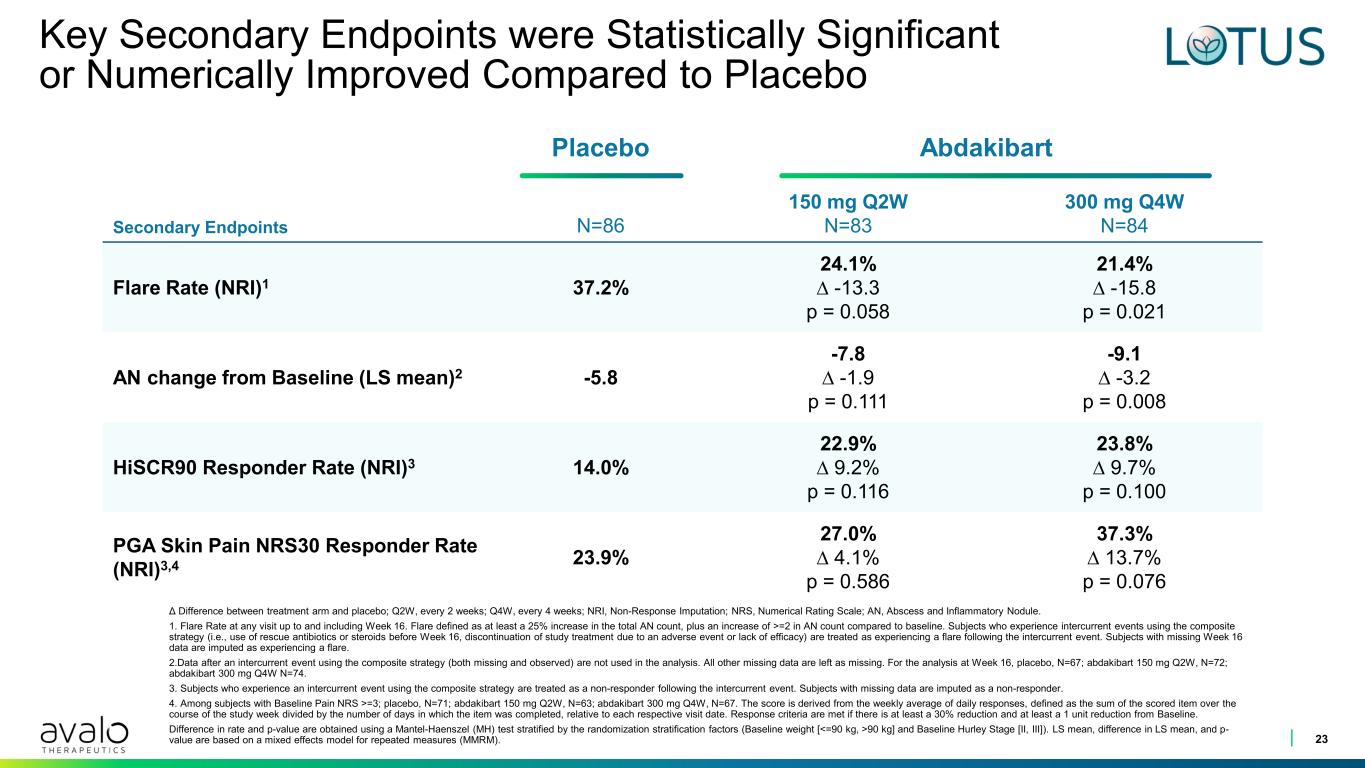
Key Secondary Endpoints were Statistically Significant or Numerically Improved Compared to Placebo 23 Δ Difference between treatment arm and placebo; Q2W, every 2 weeks; Q4W, every 4 weeks; NRI, Non-Response Imputation; NRS, Numerical Rating Scale; AN, Abscess and Inflammatory Nodule. 1. Flare Rate at any visit up to and including Week 16. Flare defined as at least a 25% increase in the total AN count, plus an increase of >=2 in AN count compared to baseline. Subjects who experience intercurrent events using the composite strategy (i.e., use of rescue antibiotics or steroids before Week 16, discontinuation of study treatment due to an adverse event or lack of efficacy) are treated as experiencing a flare following the intercurrent event. Subjects with missing Week 16 data are imputed as experiencing a flare. 2.Data after an intercurrent event using the composite strategy (both missing and observed) are not used in the analysis. All other missing data are left as missing. For the analysis at Week 16, placebo, N=67; abdakibart 150 mg Q2W, N=72; abdakibart 300 mg Q4W N=74. 3. Subjects who experience an intercurrent event using the composite strategy are treated as a non-responder following the intercurrent event. Subjects with missing data are imputed as a non-responder. 4. Among subjects with Baseline Pain NRS >=3; placebo, N=71; abdakibart 150 mg Q2W, N=63; abdakibart 300 mg Q4W, N=67. The score is derived from the weekly average of daily responses, defined as the sum of the scored item over the course of the study week divided by the number of days in which the item was completed, relative to each respective visit date. Response criteria are met if there is at least a 30% reduction and at least a 1 unit reduction from Baseline. Difference in rate and p-value are obtained using a Mantel-Haenszel (MH) test stratified by the randomization stratification factors (Baseline weight [<=90 kg, >90 kg] and Baseline Hurley Stage [II, III]). LS mean, difference in LS mean, and p- value are based on a mixed effects model for repeated measures (MMRM). Placebo Abdakibart Secondary Endpoints N=86 150 mg Q2W N=83 300 mg Q4W N=84 Flare Rate (NRI)1 37.2% 24.1% ∆ -13.3 p = 0.058 21.4% ∆ -15.8 p = 0.021 AN change from Baseline (LS mean)2 -5.8 -7.8 ∆ -1.9 p = 0.111 -9.1 ∆ -3.2 p = 0.008 HiSCR90 Responder Rate (NRI)3 14.0% 22.9% ∆ 9.2% p = 0.116 23.8% ∆ 9.7% p = 0.100 PGA Skin Pain NRS30 Responder Rate (NRI)3,4 23.9% 27.0% ∆ 4.1% p = 0.586 37.3% ∆ 13.7% p = 0.076

NASDAQ: AVTX www.avalotx.com 24